Advantages
- Enables battery-free, implantable tumor-treating field delivery directly at the tumor site
- Eliminates skull attenuation and cerebrospinal fluid shunting that limit conventional scalp-based TTF systems
- Fourfold higher ultrasonic-to-electric power conversion compared to standard cubic receivers
- Closed-loop field control maintains stable, clinically relevant electric field strength despite tissue variability
- Broad angular tolerance supports reliable transcranial ultrasonic powering
Summary
Tumor Treating Fields (TTF) therapy is clinically approved for glioblastoma but is currently delivered through external scalp arrays that must be worn for extended periods. Skull attenuation, cerebrospinal fluid shunting, and inconsistent field distribution reduce on-target dosing for deep or hard-to-reach tumors. External systems also create adherence challenges and dermatologic side effects, limiting patient comfort and long-term compliance.
This technology introduces a battery-free, ultrasonically powered implantable TTF system that delivers controlled electric fields directly at the tumor interface. A shape-engineered BaTiO₃ pyramid receiver converts focused ultrasound into localized therapeutic fields with significantly improved efficiency and angular tolerance. Integrated closed-loop sensing stabilizes field strength within clinically validated ranges, enabling precise, consistent dosing while reducing external hardware burden.
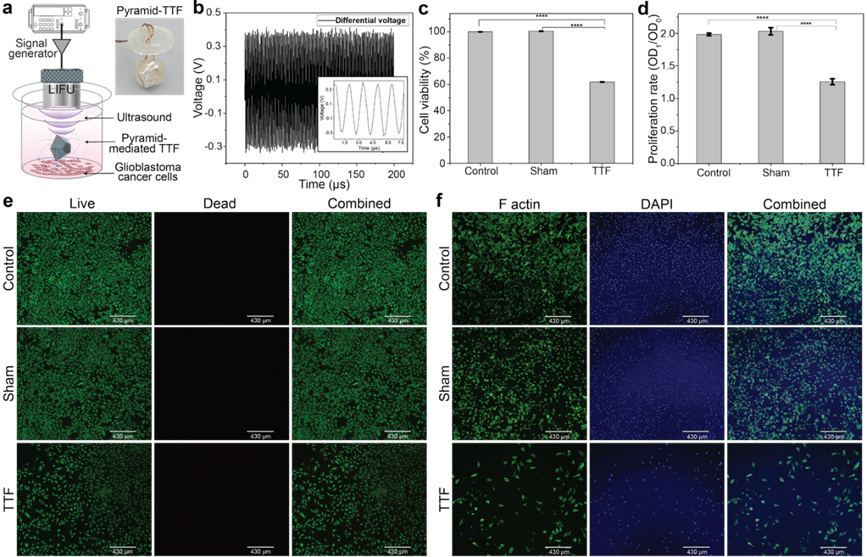
The image shows the implantable ultrasonic-powered TTF prototype and corresponding in vitro results demonstrating reduced glioblastoma cell viability after treatment.
Desired Partnerships
- License
- Sponsored Research
- Co-Development