Advantages
- Targets spore colonization, the root cause of CDI recurrence
- Broad coverage across diverse C. difficile strains
- Demonstrated protection in mice (reduced disease severity, toxins, and spore burden)
- Flexible delivery as injectable protein or oral L. lactis, enabling systemic + gut‑level immunity
Summary
Clostridioides difficile remains a leading cause of antibiotic‑associated diarrhea and pseudomembranous colitis, with treatment constrained by intrinsic antimicrobial resistance. Recurrence is driven by resilient, gut‑persistent spores that evade antibiotics and re‑seed disease. Clinically advanced vaccine programs have largely focused on toxoids (TcdA/TcdB), yet no vaccine has been approved, suggesting that anti‑toxin immunity alone may be insufficient to prevent spore‑mediated colonization and relapse.
Our inventors have developed a novel fusion immunogen, CdeCM, by combining two highly conserved, surface‑exposed C. difficile spore-surface proteins, CdeC and CdeM, to directly block the spores from colonizing the gut, which is the main driver of infection recurrence. The vaccine can be delivered either as a purified injectable protein or through a safe oral Lactococcus lactis strain engineered to produce CdeCM, enabling strong immune responses in both the bloodstream and the gut. Preclinical studies in mice demonstrate that CdeCM elicits strong serum and fecal antibody responses and delivers significant protection against challenges with hypervirulent strain, including reduced weight loss, diarrhea, toxin levels, and spore burden. Together, these results establish CdeCM as a compelling, next‑generation vaccine candidate designed to overcome the limitations of toxin‑only vaccines and meaningfully reduce CDI relapse risk.
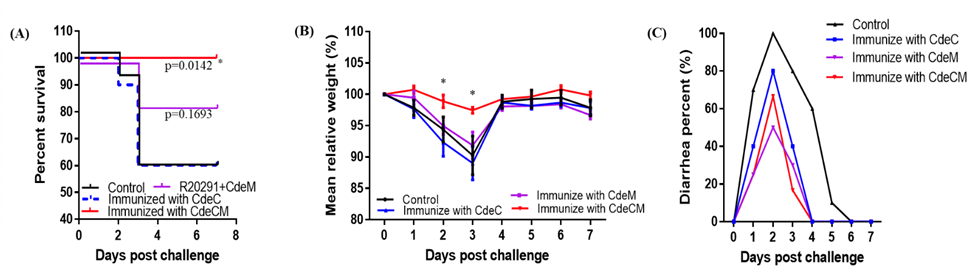
Immunizations of mice with CdeCM provide significant protection against infection with C. difficile: CdeCM‑vaccinated mice demonstrate markedly improved survival, reduced disease severity, and lower rates of weight loss and diarrhea following challenge with the hypervirulent R20291 strain.
Desired Partnerships
- License
- Sponsored Research
- Co-Development