Advantages
- Upstream pathway control: Suppresses VEGF‑A by targeting aPKC, a master regulator of angiogenic signaling.
- Selective, small-molecule mechanism: offers lower immunogenicity and easier development than antibody-based therapies.
- Combination-therapy ready: upstream angiogenic pathway control enables synergy with existing treatments.
Summary
Neuroblastoma remains a leading cause of pediatric cancer mortality, with five‑year survival hovering near 40–50% despite intensive multimodal therapy. Tumor angiogenesis—driven by hypoxia‑responsive signaling and VEGF‑A upregulation—enables growth, immune evasion, and dissemination. Existing anti‑angiogenic approaches rely heavily on monoclonal antibodies and broad‑spectrum kinase inhibitors that can be costly, immunogenic, and prone to resistance.
Our inventors have developed two complementary, small‑molecule inhibitors—ICA‑1S (a PKC‑ι–selective agent) and ζ‑Stat (a PKC‑ζ–selective agent)—that converge on inhibiting atypical PKC (aPKC) signaling to suppress the 14‑3‑3ζ / HDAC4 / HIF‑1α → VEGF‑A angiogenic axis in neuroblastoma. In pre-clinical studies using neuroblastoma cells placed under hypoxia‑like stress, treatment with either ICA‑1S or ζ‑Stat consistently reduced aPKC activity and led to lower levels of 14‑3‑3ζ, HDAC4, HIF‑1α, and ultimately VEGF‑A, indicating suppression of this angiogenic cascade. By targeting a master upstream regulatory node—aPKC‑ι/ζ—rather than VEGF‑A directly, ICA‑1S and ζ‑Stat offer a differentiated path to anti‑angiogenic control that may translate to improved efficacy, lower immunogenic burden, and combinability with current standards of care.
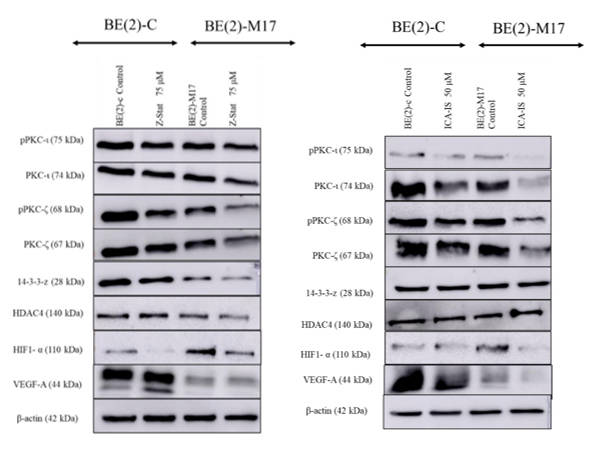
aPKC inhibition with Z-Stat (left) and ICA-1S (right)suppresses angiogenic signaling in BE(2)‑C and BE(2)‑M17 neuroblastoma cells.
Desired Partnerships
- License
- Sponsored Research
- Co-Development