Advantages:
- Permeabilizes cell membranes and tissues without applying a current minimizes the risk of ventricular fibrillation, pain, muscle contractions, and twitching
- Achieves molecular delivery and cell ablation solely through voltage application, reducing treatment complexity and potential medication side effects
- Safer modality suitable for clinical applications from gene therapy to electrochemotherapy
Summary:
Many current electroporation techniques, widely used in gene therapy and cancer treatment, face challenges because they rely on direct electrode-tissue contact that result in current flow. This flow can inadvertently stimulate excitable cells, leading to adverse effects such as pain, muscle contractions, and even life‐threatening arrhythmias like ventricular fibrillation. Additionally, balancing effective cell membrane permeabilization with the minimization of unwanted side effects remains complex, as optimizing parameters such as voltage, pulse duration, and inter-pulse intervals is crucial yet difficult to achieve without triggering significant electrical conduction through the cellular environment.
Our researchers introduce a novel electroporation method employing a dual-electrode system where one electrode directly contacts the tissue to dissipate voltage while the second, insulated with a dielectric material (dielectric constant ≥ 1), applies pulsed electric fields without creating a conductive current path. This design uniquely eliminates the current flow that traditionally causes pain, muscle twitching, and risks such as ventricular fibrillation, yet still delivers sufficient high-voltage pulses (130V to 800V) with controlled pulse durations (10µs to 100µs) and intervals to enable both reversible and irreversible membrane permeabilization. By decoupling the electric field application from direct current transmission, the invention offers a safer, more versatile approach for molecular delivery and cell ablation, thus addressing and overcoming critical limitations of conventional electroporation techniques.
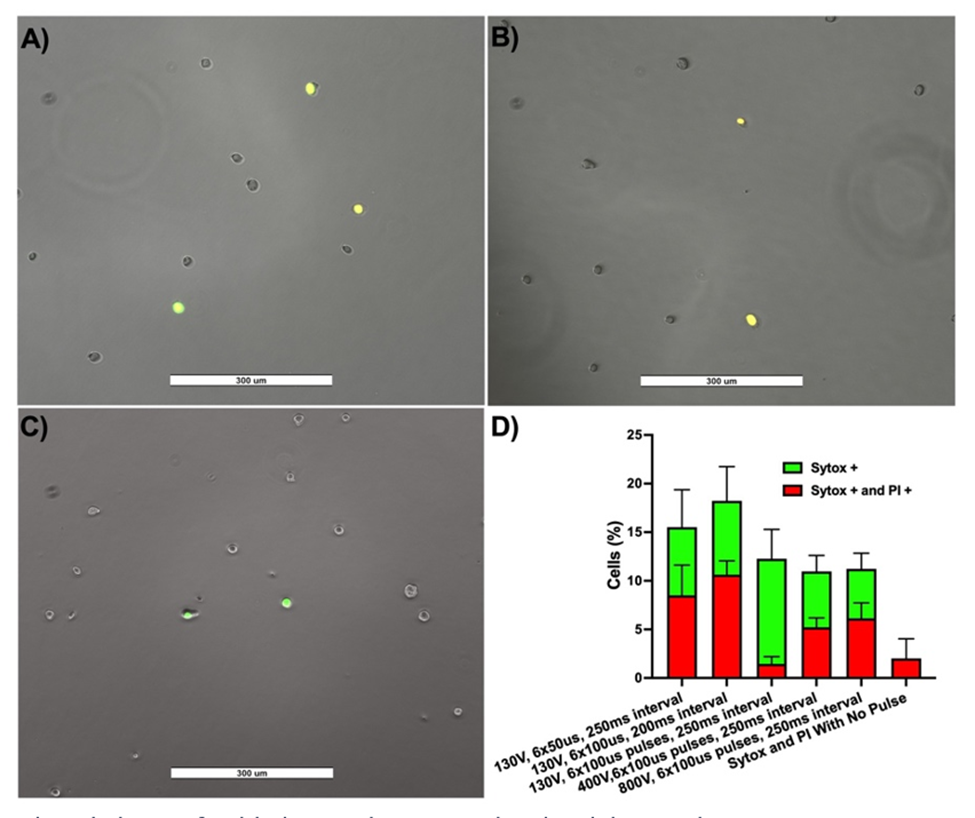
Figure-1 shows enhanced Sytox™ delivery and PI exclusion, indicating the feasibility of molecular delivery and transient electroporation induced with electric fields without current. (A), 6 pulses of 400V, 100us, and 250ms interval (B), 6 pulses of 800V, 100us, and 250ms interval (C). Five fields of view were collected for all groups and quantified (D)
Desired Partnerships:
- License
- Sponsored Research
- Co-Development