Advantages:
- ICA-1S uniquely inhibits aPKC-ι and Connexin 43, critical proteins in Glioblastoma Multiforme progression
- Significantly reduces glioblastoma cell proliferation, demonstrating superior efficacy compared to current chemotherapy agents
- Overcome therapeutic resistance by targeting Connexin 43, enhancing responsiveness to treatment
- Addressing the transcriptional and genomic diversity of GBM, ICA-1S offers broad effectiveness against cancer therapy
Summary:
Glioblastoma multiforme (GBM) is an aggressive and deadly brain tumor characterized by rapid proliferation, diffuse infiltration, and significant resistance to existing therapies, including surgery, chemotherapy, and radiotherapy. Atypical Protein Kinase C-iota (aPKC-ι) is often overexpressed in GBM and plays a crucial role in tumorigenesis by activating the PI3K/AKT/mTOR signaling pathway, which promotes cell growth and survival. Additionally, Connexin 43, a gap junction protein, contributes to GBM progression by facilitating cell-cell communication that enhances invasion and resistance mechanisms. Targeting these proteins individually has been challenging due to their complex interactions and the compensatory mechanisms of cancer cells.
Our researchers introduce a novel therapeutic approach for treating glioblastoma multiforme (GBM) by utilizing ICA-1S, a potent inhibitor of atypical Protein Kinase C-iota (aPKC-ι), to disrupt its interaction with Connexin 43 in GBM cells. The technical features involve demonstrating that ICA-1S treatment leads to a 50% reduction in T98 glioblastoma cell proliferation, downregulates both aPKC-ι and Connexin 43 proteins, and subsequently suppresses the PI3K/AKT/mTOR signaling pathway, which is vital for GBM growth and progression. This approach is novel compared to existing therapies because it specifically targets the interaction between aPKC-ι and Connexin 43—a mechanism confirmed through co-immunoprecipitation—offering a new avenue to inhibit GBM progression by affecting key proteins involved in cancer invasion and resistance that are not directly addressed by current treatments.
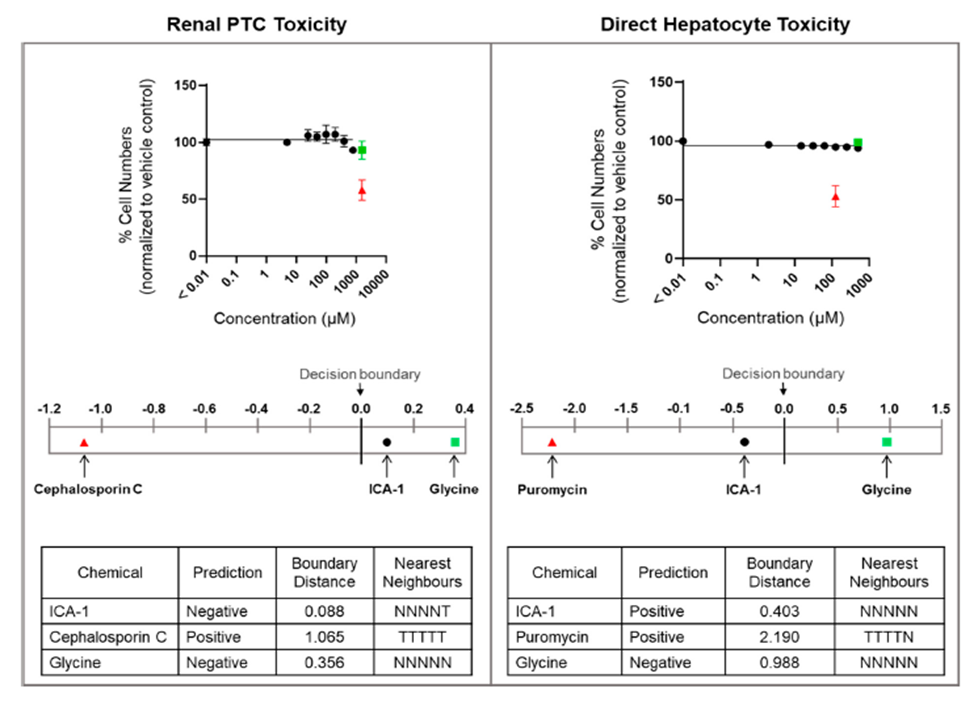 Figure-1 Analysis of ICA-1S-induced effects and toxicity prediction. The left-hand column summarizes the results relevant to the prediction of renal PTC toxicity in humans. All results were obtained using HPTCs. The right-hand column displays the results relevant for the prediction of direct hepatocyte toxicity in humans, and the results were obtained with HepaRG cells.
Figure-1 Analysis of ICA-1S-induced effects and toxicity prediction. The left-hand column summarizes the results relevant to the prediction of renal PTC toxicity in humans. All results were obtained using HPTCs. The right-hand column displays the results relevant for the prediction of direct hepatocyte toxicity in humans, and the results were obtained with HepaRG cells.
Desired Partnerships:
- License
- Sponsored Research
- Co-Development