Advantages:
- A novel method for targeting m6A methylation via YTHDF1 offers a potential therapy for tauopathies and Alzheimer’s disease
- YTHDF1 reduces neuroinflammation in Alzheimer’s disease by downregulating pro-inflammatory genes like Ccl6 and Cst7
- Enhance working memory and reduce hyperactivity in tauopathy, suggesting cognitive benefits for Alzheimer’s patients
- Offer a more efficient and cost-effective therapeutic approach
Summary:
Elucidating the significance of epitranscriptomic regulation of gene expression in memory and brain diseases is a nascent field of study. Emerging studies predict that mRNA methylation could be potentially involved in neuronal mechanisms involved in Alzheimer's disease (AD). The most abundant modification in eukaryotic mRNA and non-coding RNA is N6-methyladenosine (m6A). Nevertheless, very little is experimentally known about the impact of RNA m6A modification on AD pathophysiology. An earlier study indicates that in AD, the assembly of tau oligomers with stress granules is increased, which recruits RNA-binding proteins and the m6A transcripts and contributes to tau oligomer toxicity in the cytoplasm.
This invention targets the m6A reader protein YTHDF1 as a therapeutic strategy for Alzheimer's disease and related tauopathies. A mouse model, PS19:Ythdf1-/-, was used to test and validate the approach of targeting YTHDF1, revealing its critical role in promoting tau pathology-related immune responses. By deleting YTHDF1, the researchers observed increased soluble phosphorylated tau, decreased insoluble tau aggregates, reduced hyperactivity, improved working memory, and suppressed expression of neuroinflammatory genes such as Ccl6 and Itgax. This contrasts with existing approaches by highlighting, for the first time, the direct involvement of m6A-mediated mRNA regulation via YTHDF1 in tau-mediated neurodegeneration and neuroinflammation. Additionally, the identification of specific methylated mRNA targets of YTHDF1, like Nceh1, Egr1, and Timp2, provides novel insights into the molecular mechanisms underlying tau pathology, positioning YTHDF1 as a potential therapeutic target for treating tauopathies and Alzheimer's disease.
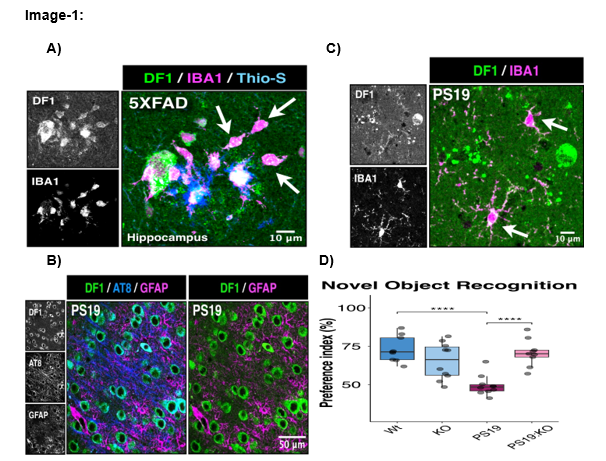
Figure 1. A) YTHDF1 in microglia surrounding Aβ deposits in the brain of 5XFAD mouse model of Alzheimer’s amyloidosis. B) Shows immunostaining of PS19 mice revealed YTHDF1 (green) and p-tau staining (blue) in AT8+ neurons. C) Shows YTHDF1 expression is observed in Iba1+microglia. D) 8 months of age, male mice (n=10 Wt, 9 KO, 10 PS19, P PS19 Ythd1-/-) were characterized by novel object recognition test. The loss of YTHDF1 rescued working memory, which is worsened due to tau pathology in PS19 mice.
Desired Partnerships:
- License
- Sponsored Research
- Co-Development