Advantages:
- Earlier diagnosis than traditional FGR screening: can detect risk as early as the 1st or 2nd trimester
- Non-invasive testing with maternal blood
- Superior sensitivity and specificity compared to ultrasound-based detection
- Can be potentially further developed into a commercial product/ standard prenatal screening test for DNA methylation for FGR screening
Summary:
FGR (Fetal Growth Restriction) is one of the most prevalent pregnancy complications, occurring in about 10% of pregnancies. Current detection methods rely heavily on ultrasound measurements of fetal weight and abdominal circumference, yet these often identify growth problems only in later stages, making early intervention challenging. There is currently no screening test available that reliably predicts the development of FGR during early pregnancy.
Our researchers have developed a novel method of testing FGR in a fetus by analyzing DNA methylation patterns in a group of genes within cell-free fetal DNA. This method achieves high sensitivity and specificity in early FGR detection through identifying differentially methylated positions (MVPs) and calculating ratios of hypo- and hypermethylated CpGs.These findings enable potential early screening in the first and second trimesters, offering a more precise tool for identifying high-risk pregnancies.
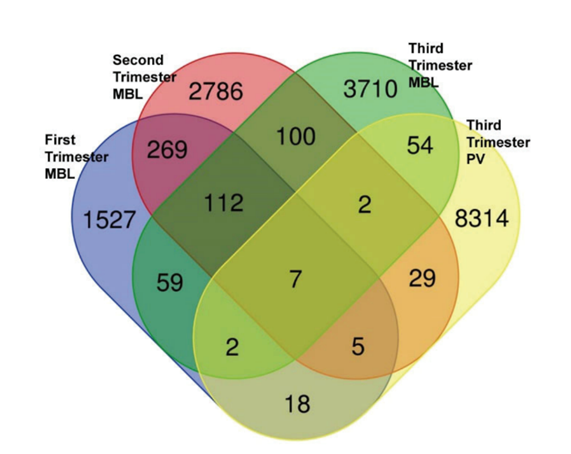
Venn diagram displaying differentially methylated CpGs in DNA MVPs in maternal blood leukocytes or placental villi in GA- and BMI-matched FGR-complicated vs. normal pregnancies. There were 7 differentially methylated CpGs detected in all specimens and 112 differentially methylated CpGs detected in the first, second, and third trimester in maternal blood leukocytes. MBL: maternal blood leukocytes; PV: Placental villi.
Desired Partnerships:
- License
- Sponsored Research
- Co-Development