Advantages:
- Asymmetric biphasic pulses significantly reduced stimulation of skeletal muscle contractions in vivo and enhanced gene delivery
- Enables delivery of insulin and glucokinase to replace daily injections with long-term, non-immunogenic protein expression in skeletal muscle
- Higher-efficiency gene therapies avoid immunogenicity and off-target integration, offering a safer, cost-effective solution
- Portable, clinician-friendly device design improves clinical adoption and accessibility
Summary:
Existing gene therapy strategies encounter significant obstacles that hinder broad clinical use. Non-viral electrotransfer techniques often suffer from low transfection efficiency and induce problematic muscle contractions, whereas viral methods pose immunogenicity concerns, possible off-target genetic integration, and high production costs. Such limitations are especially pronounced for conditions that require long-term protein supplementation, including metabolic disorders and coagulation deficiencies, where repeated administration or immunosuppression is burdensome. Together, these challenges underscore the need for an efficient, non-immunogenic, and affordable approach that delivers stable gene expression without major side effects.
Our researchers have developed a method to address the issue of muscle contractions. The invention utilizes a different form of electric pulses and demonstrates that asymmetric, biphasic pulse sequences can enhance gene delivery to tissues while reducing the stimulation of skeletal muscle, thereby minimizing contractions. This platform employs localized in vivo electrogene therapy (liveGT) with specially designed asymmetric, biphasic pulses under two microseconds to deliver plasmid DNA into skeletal muscle without triggering muscle contractions. Operating at frequencies around 100 kHz and using carefully controlled pulse sequences, it achieves efficient DNA uptake while avoiding the immunomodulatory complications and off-target risks associated with viral methods. A handheld, battery-powered system with H-bridge circuitry and GaNFETs precisely administers these pulses and can be programmed to meet different therapeutic needs, as demonstrated in rat and pig models. The device’s portable design, non-stimulating waveforms, and ability to co-deliver multiple genes make it suitable for broad therapeutic applications, exemplified by its potential to replace frequent insulin injections in Type 1 Diabetes with stable gene expression over extended periods.
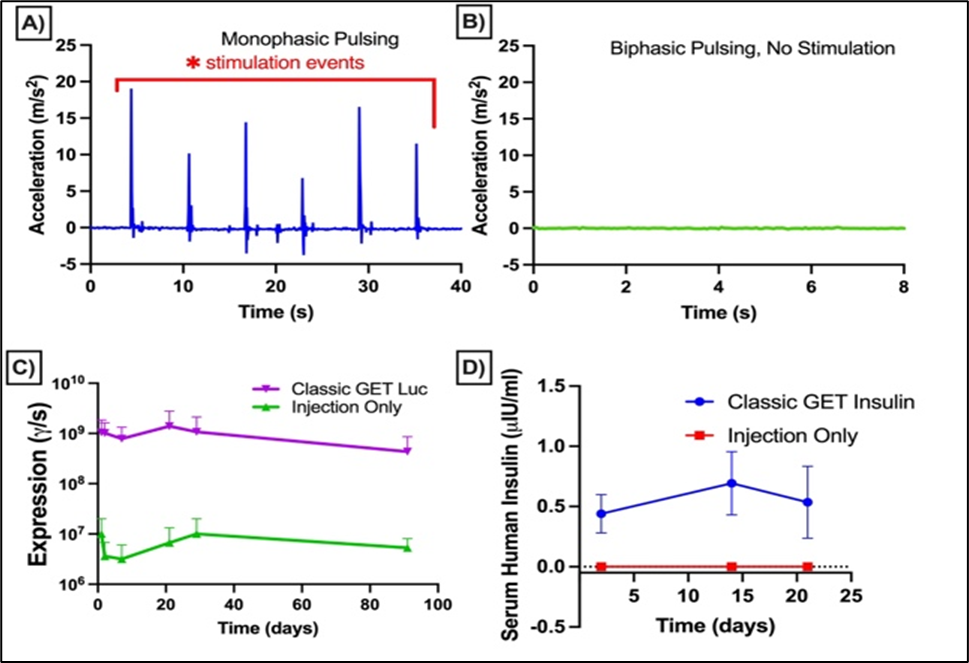
Image Description: Asymmetric, biphasic pulses are non-stimulating in a Guinea Pig model of gene delivery to the skin (A-B). Localized expression in skeletal muscle can be maintained long-term (C). Skeletal muscle can secrete human insulin into the blood stream (D).
Desired Partnerships:
- License
- Sponsored Research
- Co-Development