Advantages:
- Repurposes an existing FDA-approved drug
- Achieves dose-dependent inhibition of ASPC-1 pancreatic cancer cell proliferation
- May enhance treatment efficacy when combined with standard chemotherapy
Summary:
Pancreatic cancer remains one of the most lethal malignancies due to late diagnosis, limited treatment efficacy, and a tumor microenvironment that hinders drug delivery. Traditional therapies, including surgery and chemotherapy, offer only modest survival benefits and often cause toxic side effects, while immunotherapies have shown limited success due to immune suppression. There is a critical need for new treatment strategies that overcome drug resistance, reduce toxicity, and improve patient outcomes.
Our researchers have discovered that the FDA-approved HIV medication Stavudine can be repurposed as a novel potential anticancer agent by targeting atypical protein kinase C (aPKC) signaling pathways. In ASPC-1 pancreatic cancer cells, Stavudine demonstrated a significant, dose-dependent inhibition of cell proliferation, with immunoprecipitation studies revealing a distinct mechanism not seen in conventional treatments. Stavudine carries the potential to be repurposed for hard-to-cure cancerous diseases, cancerous diseases such as pancreatic cancer.
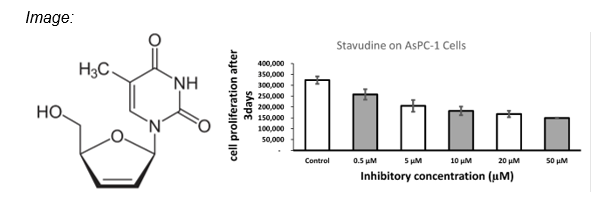
Preclinical data from ASPC-1 pancreatic cancer cells shows a decreasing trend in cancer cell viability with increasing Stavudine concentrations, highlighting its potential therapeutic effect.
Desired Partnerships:
- License
- Sponsored Research
- Co-Development