Advantages:
- Bivalirudin-tagged nanoparticles bind and neutralize nascent thrombin in clots, minimizing systemic anticoagulant effects
- Anti-thrombin nanoparticle confines its anticoagulant activity to sites of clot formation, limiting excessive systemic coagulation inhibition
- Dual therapeutic and diagnostic potential allows clinicians to treat NEC and monitor thrombin-driven injury in real-time
- Anti-thrombin nanoparticles can be administered after disease onset, reducing intestinal damage and inflammatory markers
Summary:
Necrotizing enterocolitis (NEC) primarily affects premature infants and is characterized by an inflammatory necrosis of the bowel often accompanied by thrombocytopenia, prompting frequent platelet transfusions. However, these standard interventions may overlook the underlying mechanisms that drive early platelet activation and inflammation, particularly the role of thrombin generation and tissue factor release in a setting where neonates have a developmental deficiency of natural anticoagulants. Existing therapies focus on managing symptoms and balancing blood product transfusions while avoiding complications like bleeding, yet they can fail to address the rapid progression of NEC and the critical timing of platelet activation events. This leaves a pressing need for approaches that more precisely mitigate both tissue injury and heightened inflammatory responses without exacerbating hemorrhagic risk.
A neonatal murine model is employed to replicate key aspects of necrotizing enterocolitis (NEC) by administering trinitrobenzene sulfonate (TNBS) in 10-day-old mice, revealing rapid platelet activation triggered by pronounced thrombin activity. Resident intestinal macrophages release tissue factor, causing excessive thrombin generation in neonates due to naturally low levels of antithrombin and α2-macroglobulin. This early platelet activation precedes overt intestinal damage and is effectively targeted by bivalirudin-tagged perfluorocarbon nanoparticles, which bind nascent thrombin in clots. Administered after NEC onset, these nanoparticles mitigate intestinal injury and inflammatory markers without increasing hemorrhage risk, illustrating a targeted approach to controlling thrombin-driven platelet activation in NEC.
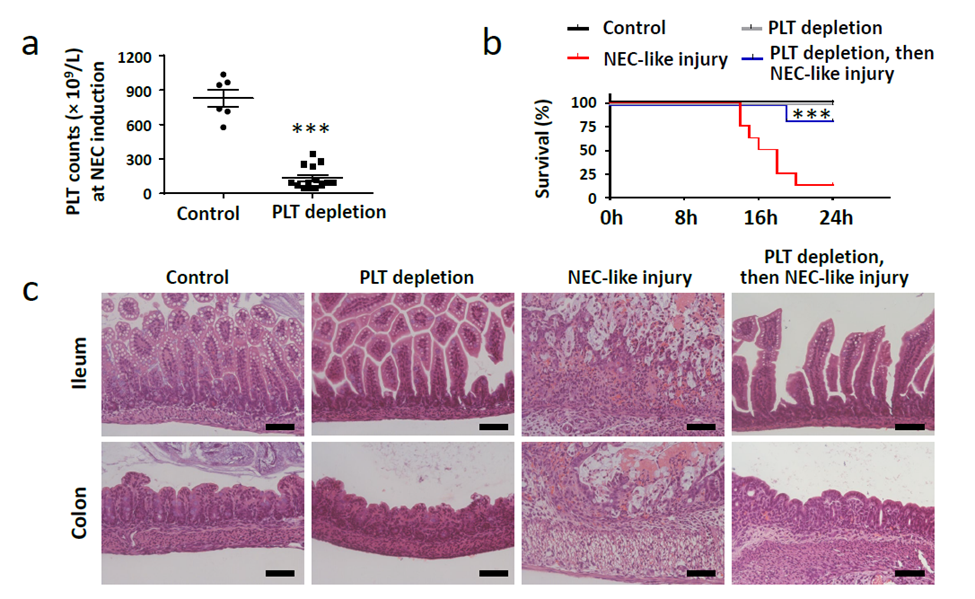
A) Depleted platelet counts to 50-100 × 109/L, levels seen during moderate-severe murine NEC-like intestinal injury, B & C) Platelet depletion reduced mortality and the severity of intestinal injury.
Desired Partnership: