Advantages:
- Uniquely replicates the clinical scenario of RBC transfusion-exacerbated NEC in anemic neonates, providing a more accurate model than the traditional method
- Provide superior mechanistic insights, offering precise investigation of molecular pathways compared to in vitro intestinal injury assays
- Cost-effective and high-throughput experimentation, facilitating faster experimental cycles and enabling robust statistical power at lower overall cost
- Ability to manipulate genetic and immune factors by detailed dissection of transfusion-triggered immune responses and inflammation
Summary:
Necrotizing enterocolitis (NEC) is a severe inflammatory disease of the intestine that disproportionately affects premature infants and is linked to significant morbidity and mortality. Multiple retrospective clinical studies have reported an association between red blood cell (RBC) transfusions and NEC, yet questions of causality remain unresolved due to observational study designs and the challenges of controlling for confounding factors. Existing investigations have been hampered by varying patient populations, differences in clinical management, and the lack of a rigorous mechanism-based approach to establish whether anemia, transfusion, or both collectively lead to bowel injury. As a result, clinicians are frequently faced with difficult decisions about transfusion thresholds and timing without definitive guidance on how to minimize the risk of NEC while ensuring adequate treatment of neonatal anemia.
Researchers developed a murine model showing that RBC transfusions can trigger necrotizing enterocolitis (NEC) in severely anemic neonatal mice, focusing on a hypoxia-reoxygenation mechanism. Severe anemia was induced through repeated phlebotomy, followed by transfusion of leukoreduced RBCs on day 11. Only anemic-transfused pups developed NEC-like lesions centered in the ileocecal and mid-colonic segments, exhibiting coagulation necrosis, hemorrhage, inflammation, and elevated gut permeability. Mechanistic studies indicated that these injuries resulted from macrophage-driven inflammation rather than ischemia: macrophages showed increased activation (evidenced by elevated F4/80hi, NF-κB signaling, and ROS production), while depleting macrophages prevented injury. Anemia alone created a proinflammatory intestinal environment, which RBC transfusions significantly amplified, and results further showed that washing stored RBCs reduced disease severity. This model recapitulates key pathological and molecular features of human NEC and highlights the role of pre-existing gut inflammation, the severity of anemia, and the importance of refined transfusion practices in preventing NEC-like injury.
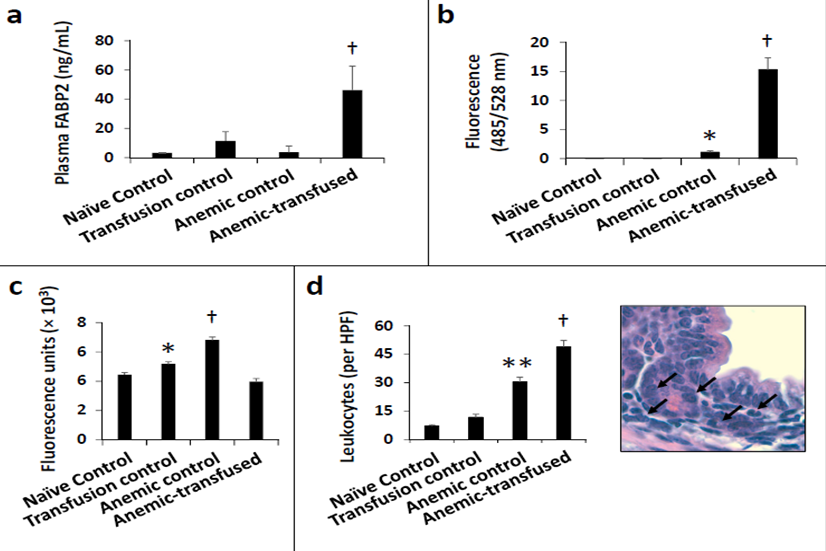
A) The anemic-transfused groups showed a 4-fold elevation of FABP2 levels compared to control (p<0.001), B) The fluorescence intensity was 0.01 ± 0, 0.05 ± 0.01, 1.07 ± 0.26, and 11.51 ± 1.76 units/mL in the naïve control, transfusion control, anemic control, and anemic-transfused groups, respectively, C) The anemic-transfused group showed regional blood flow that was similar to naïve controls, and was not suppressed below normal D) increased cellularity in NEC lesions in the anemic-transfused group; many infiltrating cells showed a macrophage-like appearance (large, rounded cells with eccentrically placed vesicular, reniform nuclei.
Desired Partnerships:
- License
- Co-Development
- Sponsored Research